The Elbow
A CONSIDERATION OF THE ELBOW AS A TENSEGRITY STRUCTURE
International Journal of Osteopathic Medicine
2012;15:53-65. link
Graham Scarr request copy
ABSTRACT
The elbow is conventionally described as a uniaxial hinge joint and the pivot of proximal forearm rotation; the joint surfaces guide motion, the ligaments maintain joint integrity and the muscles cause motion. However, this simplicity is less clear on detailed examination and masks uncertainties over its component structures and their functions.
Elbow anatomy is examined from a tensegrity perspective with a re-assessment of these functions. Tensegrity structures, like the elbow, are inherently stable and maintain a balanced equilibrium during changes in shape because of ‘continuous tension’. Connective tissues mechanically integrate local and distantly related components into a single functional unit while proprioceptive sensors neurally influence motor activity; both control joint dynamics.
It is suggested that this has relevance to understanding the commonly encountered but vague pathologies such as ‘tennis elbow’ and ‘repetitive strain injury’; the aetiologies of these conditions continue to be the subject of debate.
INTRODUCTION
The human elbow is commonly described as a uniaxial hinge joint that permits movement in a single plane and the pivot of proximal forearm rotation. It enables the length of the upper limb to vary through 145o of flexion and the forearm to rotate 150o thus facilitating the hand in carrying out its functions of grasping and manipulation. However, this simplicity is less clear on detailed examination and masks uncertainties over its component structures and their functions. In particular, the relationship between adjacent joint surfaces, variations in ligamentous complexes and the precise role of certain muscles; movement analysis frequently overlooks the variable fulcrum1 and helicoid motion.2 Elbow anatomy is examined from a tensegrity perspective with re-assessment of its component functions and their integration into a global structural system. It is suggested that this has relevance in understanding the commonly encountered but vague pathologies such as ‘tennis elbow’ and ‘repetitive strain injury’ (RSI).
The elbow is a common feature of the vertebrate tetrapod, but shows considerable phylogenetic variation where it is modified for crawling, speed, manipulation and flight. Smaller mammals, such as shrews (Soricidae) and squirrels (Sciuridae), maintain short, flexed forelimbs for bounding motion; whereas the elbow of the horse (Equus) is relatively immobile, with long extended limbs that give it a mechanical advantage.3 Mammalian carnivores show distinct elbow morphologies related to mutually exclusive feeding strategies. ‘Ambushers’ such as cats ( Felidae) are able to manipulate their prey because of a strong supinatory ability and have a relatively wide elbow joint. The ‘pursuers’ such as hyenas ( Hyenidae) and dogs (Canidae) have narrow box-like joints more adapted for speed. Selective influences have allowed the cheetah (Acinonyx jubatus) to display features of both groups.4 Similar differences in the macaque monkey (Macaca) distinguish tree-dwelling species adapted for power and complex rotational movements from those more suited to the speed of terrestrial locomotion.5 Although the human elbow might be able to match some of these feats its evolutionary advantage seems to be in supporting the precision of hand and finger movements.
In the discourse to follow the tensegrity concept is introduced and basic human anatomy reviewed with some particularly significant features described in detail. A discussion of tensegrity research and its relevance to biological structure and manual treatment is then given.
TENSEGRITY
The concept of tensegrity as a new structural principle started in the 1940’s with Kenneth Snelson,6 who explored and developed it through sculpture, and Buckminster Fuller7 who considered it as part of a wider system of geometry. The application to living organisms began with Ingber et al. (1981),8 while investigating the role of the cellular cytoskeleton in angiogenesis, and Levin (1982)9 who considered it from first principles and an evolutionary perspective.
 Tensegrity (tension-integrity) is popularly recognised for its distinct compression elements (struts) that appear to float within a tensioned network (cables). Because every part influences every other part, the entire system balances in a position of stable equilibrium, irrespective of the direction of gravity or a change in shape (fig. 1). This contrasts with the orthodox view of bio-mechanics that describes the musculo-skeletal system through classical Newtonian mechanics using pillars, arches and levers to counteract the force of gravity; in this approach bones are restrained by soft tissues that permit movement in a local piecemeal like way. This is a system common to man-made structures but no longer fits with biology.9,10
Tensegrity (tension-integrity) is popularly recognised for its distinct compression elements (struts) that appear to float within a tensioned network (cables). Because every part influences every other part, the entire system balances in a position of stable equilibrium, irrespective of the direction of gravity or a change in shape (fig. 1). This contrasts with the orthodox view of bio-mechanics that describes the musculo-skeletal system through classical Newtonian mechanics using pillars, arches and levers to counteract the force of gravity; in this approach bones are restrained by soft tissues that permit movement in a local piecemeal like way. This is a system common to man-made structures but no longer fits with biology.9,10
A particular feature of tensegrity models is their compression struts that do not touch each other at any point; there are no levers, no fixed fulcrums and no bending moments.7,9 Considering bones in the same way implies that the joints between them have similar characteristics but this has been contentious as anatomy texts assume joint surfaces are compressed together.2
Levin11 visually examined normal bones during surgery and found that they never compressed each other in living subjects. This would imply that bones are held apart by soft tissues and the mechanics of the shoulder,12 spine,13 pelvis,14 cranial vault15 and distal radio-ulnar joint16 have been described in this way, but these observations have not been widely recognized.
Rikli et al (2007)17 measured significant pressures across the radio-ulna-carpal joint in-vivo that contradicts the previous findings although the substantial thickness of the sensor (1.5 mm) between the joint surfaces may have influenced these results. Marai et al (2006)18 used in-vivo computerized tomography and kinematic data to demonstrate contact between carpal bones throughout movement but also suggested that unloaded carpal cartilage is minimally compressed during motion. Because lubricated synovial joints are virtually frictionless, appositional surfaces must be held in relation to each other by tensional forces on either side of the bones, and in the traditional bio-mechanics model this would increase compression.12 Baeyens et al (2006)19 used similar techniques to describe motion of the proximal and distal radio-ulnar joints but did not map the areas of contact. Taken together these findings suggest that a lot is still unknown about synovial joints.
Conventionally, the elbow is described as a hinge with the joint surfaces guiding motion, the ligaments maintaining joint integrity and the muscles causing motion, but examining the structure suggests that such neat division of function is inconsistent with the evidence.
BASIC ANATOMY
 The human elbow is made up of three joints – the humeral trochlea and ulnar trochlear notch, humeral capitulum and radial head, and the radial head with the radial notch of the ulna. The trochlea has an hourglass shape, with a medial flange that projects to a lower level and tilts the plane of the joint, and a central groove that spirals obliquely around it. The trochlear notch of the ulna separates the olecranon and coronoid processes and is divided into medial and lateral parts by a sagittal ridge matching the groove on the trochlea. The end of the radial head is concave to match the convexity of the capitulum and the radial notch on the ulna accepts the oval rim of the radial head. All three joints are enclosed in a continuous capsule lined with synovial membrane.2
The human elbow is made up of three joints – the humeral trochlea and ulnar trochlear notch, humeral capitulum and radial head, and the radial head with the radial notch of the ulna. The trochlea has an hourglass shape, with a medial flange that projects to a lower level and tilts the plane of the joint, and a central groove that spirals obliquely around it. The trochlear notch of the ulna separates the olecranon and coronoid processes and is divided into medial and lateral parts by a sagittal ridge matching the groove on the trochlea. The end of the radial head is concave to match the convexity of the capitulum and the radial notch on the ulna accepts the oval rim of the radial head. All three joints are enclosed in a continuous capsule lined with synovial membrane.2
The axis of humero-ulna motion is described as the centre of three circles formed by the edges of the condyles, the sagittal groove and medial lip of the trochlear; it is on a vertical line drawn down from the anterior cortex of the humeral shaft.
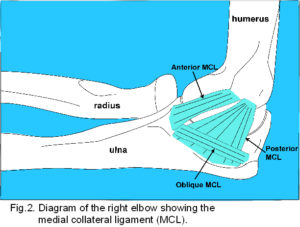
 Passive stability is maintained by ligaments. The medial collateral ligament (MCL) (fig. 2) consists of well-defined fibres originating from the anterior aspect and tip of the medial epicondyle, attaching to the proximal tubercle on the medial aspect of the coronoid process, a poorly defined thickening of the capsule from the lower posterior aspect of the medial epicondyle to the medial margin of the olecranon, and inferior oblique fibres which thicken the capsule between the olecranon and coronoid processes.2
Passive stability is maintained by ligaments. The medial collateral ligament (MCL) (fig. 2) consists of well-defined fibres originating from the anterior aspect and tip of the medial epicondyle, attaching to the proximal tubercle on the medial aspect of the coronoid process, a poorly defined thickening of the capsule from the lower posterior aspect of the medial epicondyle to the medial margin of the olecranon, and inferior oblique fibres which thicken the capsule between the olecranon and coronoid processes.2
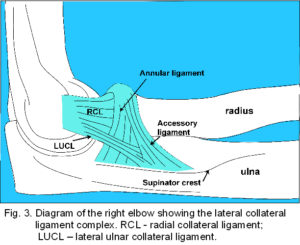
The lateral collateral ligament (LCL) (fig. 3) has a highly variable structure that includes the radial collateral ligament (RCL), lateral ulna collateral ligament (LUCL) and annular ligament.20-24 The RCL is a poorly demarcated structure extending from the infero-medial aspect of the lateral epicondyle to the margin of the radial notch, radial head22,24 and superficial lateral portion of the annular ligament.24 The LUCL extends from the lateral epicondyle to attach to a tubercle on the supinator crest of the ulna, some fibres blending with distal fibres from the annular ligament. The fibrous ring of the annular ligament encircles 4/5 of the radial head and attaches to the anterior and posterior margins of the radial notch; proximally it blends with the joint capsule2 and distally (sometimes called the accessory ligament)20,24 with the LUCL. This simple description continues with the active tensors – the muscles that cause motion (fig. 4).
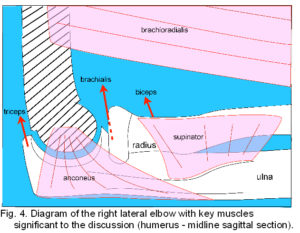 Principal flexors are biceps brachii, brachialis and brachioradialis. The two heads of biceps unite in a common tendon to attach on the radial tuberosity, brachialis attaches to a tuberosity and anterior coronoid process of the ulna, and brachioradialis attaches to the proximal 2/3 of the lateral supracondylar ridge and intermuscular septum, inserting on the radius proximal to the lateral part of the styloid process.2 Extension occurs through contraction of the three heads of triceps that unite in a common tendon attached to the upper olecranon process. Supinator rotates the radius so that the palm faces anteriorly and originates from the lateral epicondyle, RCL and annular ligament, supinator crest of the ulna (figs. 3; 4) and an aponeurosis covering the muscle, it attaches to the lateral surface of the proximal radius. Pronator teres arises as a superficial head proximal to the medial epicondyle, the common flexor tendon, the intermuscular septum between it and flexor carpi radialis, and the antebrachial fascia. An ulnar head arises from the medial coronoid process and both unite to attach to the lateral radius. Other muscles that originate from the distal humerus and attach to the wrist, hand and fingers also contribute to elbow movement but will not be detailed here.2
Principal flexors are biceps brachii, brachialis and brachioradialis. The two heads of biceps unite in a common tendon to attach on the radial tuberosity, brachialis attaches to a tuberosity and anterior coronoid process of the ulna, and brachioradialis attaches to the proximal 2/3 of the lateral supracondylar ridge and intermuscular septum, inserting on the radius proximal to the lateral part of the styloid process.2 Extension occurs through contraction of the three heads of triceps that unite in a common tendon attached to the upper olecranon process. Supinator rotates the radius so that the palm faces anteriorly and originates from the lateral epicondyle, RCL and annular ligament, supinator crest of the ulna (figs. 3; 4) and an aponeurosis covering the muscle, it attaches to the lateral surface of the proximal radius. Pronator teres arises as a superficial head proximal to the medial epicondyle, the common flexor tendon, the intermuscular septum between it and flexor carpi radialis, and the antebrachial fascia. An ulnar head arises from the medial coronoid process and both unite to attach to the lateral radius. Other muscles that originate from the distal humerus and attach to the wrist, hand and fingers also contribute to elbow movement but will not be detailed here.2
Four main nerves cross the elbow to innervate muscles and articular structures. The musculocutaneous nerve supplies biceps and brachialis proximal to the elbow with a branch innervating the anterior part of the joint capsule. The radial nerve sends medial and posterior branches to supply triceps, anconeus and the posterior and antero-lateral capsule; a lateral branch pierces the lateral intermuscular septum to supply brachialis (in part), brachioradialis and extensor carpi radialis longus before continuing as the posterior interosseous nerve to supply supinator and the wrist/hand extensors. The median nerve passes between brachialis and the bicipital aponeurosis to supply the pronator muscles and most of the wrist and hand flexors. The ulna nerve innervates the MCL and supplies flexor carpi ulnaris. Articular nerves accompany blood vessels supplying synovial membrane and fat pads and contain vasomotor fibres as well as afferents for pain and proprioception.2
A BIT MORE DETAIL
Joint surfaces
Although contours of the trochlea and trochlear notch appear to match (fig. 4), their surfaces have been shown to be incongruent, with a central space up to 2.8 mm over the sagittal ridge and 4.0 mm at the medial notch edge.25 Cartilage covering the joint surfaces increases in thickness towards the proximal and distal aspects of the trochlear notch where it accepts more load in all positions.26 The actual pattern of contact is subject to great variation between individuals, with a contact area of 8-37% for a 25 Newton (N) load, and 33-77% at 500 N loading (500 N is equivalent to 150% of the load generated by a slow two-handed push-up).25 Contact changes throughout movement and is greatest in the middle of the movement range, but these findings are based on cadaveric studies with externally and passively applied loads, they may be different in-vivo. The radial head does not make contact with the capitulum at any point during movement.21
A closer examination of the ligamentous structures also reveals some interesting details.21-24,27 The MCL attaches to the medial epicondyle just postero-inferior to the presumed axis of rotation and inserts along the trochlear notch coexistent with fibres of the capsule (fig. 2). Both the anterior and posterior MCL have distinguishable fibres that tighten in an ordered sequence from anterior to posterior during flexion, which suggests that the joint acts like a cam, at least in cadavers.23,24
The LCL (fig. 3) is really a complex of ligaments that blend into each other and there is considerable variation.20-24 The RCL attaches to the lateral epicondyle coincident with the axis of flexion although anterior and posterior fibres have a similar tension sequence to the MCL.23 Its supero-medial aspect blends with the anterior capsule, inferior border with the inferior capsule, and superficial surface with the fascia around supinator and muscles originating from the common extensor tendon.2,23,24 The LUCL varies from a thickened structure inseparable from the RCL through to a vestigial fibrous or thin inter-muscular septum between supinator and anconeus. The proportion of each type varies greatly between ethnic populations21 and its fibres have been found to be taut only above 120o flexion.23 The RCL and LUCL are not distinguished in some anatomy texts.2
The annular ligament consists of deep capsular, intermediate ligamentous and superficial fibres that can blend with the LUCL with some fibres attaching to the radial head, neck and RCL.22,24 The anterior portion is taut in supination and the posterior in pronation.20
Stability between the ulna and radius is also maintained by several other connective tissue structures. The quadrate ligament reinforces the joint capsule and limits radial rotation. Fibres of the oblique cord pass from the ulna to the radius distally and are taut in supination,28 but its functional significance has been described as “dubious”.2,20 The interosseous membrane has long broad attachments with a stout central band and several accessory bands passing obliquely from the radius to the ulna distally; they are assumed to act as restraints to proximal migration of the radial head when the forearm is loaded axially.29 An inconstant proximal band has fibres perpendicular to the central band, and membranous portions extend to the distal radio-ulnar joint capsule and articular disc. The interosseous membrane is only taut midway between full pronation and supination but the varying fibre orientations suggest more complex functions.2
Two elbow muscles are conspicuous in that their functions are poorly understood and they don’t fit neatly into the basic flexor/extensor or pronator/supinator categories (fig. 4). Brachioradialis is minimally active in slow easy flexion even though its high attachment on the supracondylar ridge gives it a mechanical advantage over the other flexors. Because brachioradialis is considered an ‘assistant’ synergist,30 its distal attachment is sometimes surgically repositioned to replace the loss of other muscles after injury;31,32 a more significant function is described later in relation to the tensegrity concept. The other muscle is anconeus, which originates from the posterior aspect of the lateral epicondyle and RCL,21 inserting into the posterior olecranon process and adjacent ulna. Anconeus is active throughout all movements of the elbow,33,34 may abduct the ulna,35 and rather vaguely is considered to have a ‘stabilizing’ function.35,36
The elbow muscles are further interesting from a tensegrity perspective because they have fibrous and fascial connections to other structures that seem unrelated to their presumed main function. The biceps tendon sends an expansion into the antebrachial fascia as the bicipital aponeurosis (lacertus fibrosus) with longitudinal and oblique fibres that create origins for the flexor carpi radialis and brachioradialis muscles.37,38 A third head of biceps can arise from the supero-medial part of brachialis to attach separately to the tendon and aponeurosis.2 Brachialis may be divided, fuse with brachioradialis, biceps or pronator teres and also attach to the bicipital aponeurosis; it sends numerous fibres to attach to the anterior capsule. Accessory slips to pronator teres may also arise from biceps and the medial intermuscular septum.2 Triceps sends fibres from its deep surface to blend with the posterior joint capsule and a fibrous expansion over muscles originating from the lateral epicondyle connects to the deep antebrachial fascia.2,37 Muscles taking origin from the common extensor tendon such as extensor carpi ulnaris, extensor digitorum and extensor digiti minimi also have numerous attachments into this expansion,2,37 while extensor carpi radialis longus and brevis connect to the lateral intermuscular septum and RCL, respectively.2
Intriguingly, anconeus is the only elbow muscle that is not attached directly to the overlying fascia;2 approaching the elbow from a different perspective may serve to better understand these structures.
 THE TENSEGRITY ELBOW
THE TENSEGRITY ELBOW
A respectable function
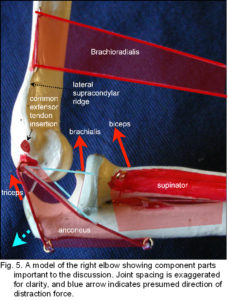
Brachioradialis differs from the other flexors in that its muscle bulk is mostly distal to the elbow (figs. 4-6). Muscles that are placed proximally reduce inertia of the limb during rapid movements ??yet brachioradialis is mainly active in rapid/forced flexion and extension;2 rather bizarrely it is supplied by the ‘extensor’ nerve (radial) that innervates triceps and anconeus, its supposed antagonists. Several investigators have shown that the elbow flexors are not a single functioning unit,33,39-41 and all these factors suggest that the principle function of brachioradialis is somewhat different.
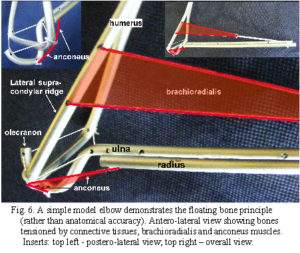
Due to the relatively high attachment on the lateral supracondylar ridge and insertion on the distal radius, brachioradialis is likely to create a distraction force between the humerus and ulna (fig. 5). As this would be contained by the tendon of triceps and collateral ligaments on both sides, the muscle acts in a trans-articular or shunt capacity, and pulls the coronoid surface towards the trochlea (the radius exerts tension on the interosseous ligament which pulls the ulna proximally) (fig. 6).2 Anconeus is also in a position to counter joint distraction, although it has different force vectors to the ligaments (figs. 4; 5), it is active throughout all movements of the elbow33,35,36 and always precedes triceps activity.34 Over about 90o of flexion, biceps, brachialis and triceps will tend to pull the olecranon surface towards the trochlea.
Do brachioradialis and anconeus, therefore, actively balance pressures exerted at the joint surfaces in a controlled way? Is it even possible for the surfaces to lose contact with each other during movement?
Discontinuous compression
 Conventionally, synovial joints are considered to transfer compressional forces across their surfaces and it is expected that the effects of this would be apparent in the underlying bone. However, although a greater density of subchondral bone does occur beneath proximal and distal aspects of the notch, it has been found to reflect overall stresses imposed on the ulna rather than local pressure on the joint surfaces.26,41,42 Eckstein et al (1995)26 noted that positioning the joint reaction force close to the centre of the socket through neuromuscular control would balance any compression stresses. It is possible that joint surfaces maintain contact by just sliding over each other, rather than compressing, with brachioradialis and anconeus regulating the pressure across the humero-ulnar joint.
Conventionally, synovial joints are considered to transfer compressional forces across their surfaces and it is expected that the effects of this would be apparent in the underlying bone. However, although a greater density of subchondral bone does occur beneath proximal and distal aspects of the notch, it has been found to reflect overall stresses imposed on the ulna rather than local pressure on the joint surfaces.26,41,42 Eckstein et al (1995)26 noted that positioning the joint reaction force close to the centre of the socket through neuromuscular control would balance any compression stresses. It is possible that joint surfaces maintain contact by just sliding over each other, rather than compressing, with brachioradialis and anconeus regulating the pressure across the humero-ulnar joint.
In cadavers, incongruity between the trochlea and trochlear notch causes contact area to vary with joint position and load, with the entire joint surface encountering some mechanical stimulation during cyclic loading.1,25,26 If this occurred in-vivo it would significantly reduce the peak stresses acting on articular surfaces as well as improve cartilage lubrication and chondrocyte activity.26,42Although one of the principles of tensegrity is that compression is discontinuous,7 i.e. it is not transferred directly between struts (bones), sliding surfaces are not inconsistent with this in a biological context.13,16 It may be that normal cartilage functions as a protective cushion rather than major load bearer, with tensioned structures regulating joint spacing and/or surface pressure; the mechanical properties of cartilage are still not fully understood. (Since this was accepted for publication, a recent paper reviews the evidence for meniscal cartilage compression in the knee joint, and supports the thesis that cartilage does not normally play a major compressive role: Andrews S, Shrive N, Ronsky J. The shocking truth about meniscus. J Biomechanics 2011;44:2737-40.) link
Bones, the largest struts in the system, are compressed by muscles and fascia under tension but it would be the tensegrity configuration that holds them apart. Sverdlova and Witzel (2010)43 showed how the balance of agonist and antagonist muscle tensions reduces stress concentrations in long bones and make them compatible with the resiliency required of tensegrity struts. In addition, the diameters of certain muscles increase with axial contraction, making them variable length compression struts that are likely to change the tension in associated fascia; in fact, a strut could be considered as any structure that maintains a separation, such as across bones, muscles or even blood vessels.
Continuous tension
Although brachioradialis and anconeus have been highlighted as possible joint space/pressure modulators it is likely that multiple muscles and connective tissues would contribute to this. It is generally assumed that tensional forces cross synovial joints by means of a parallel system of active muscle and passive ligaments, as already described, but newer research suggests otherwise.
Things are not what they seem
Ligaments are conventionally described as guide wires or restrainers of excess motion with the MCL and RCL resisting valgus and varus stresses, respectively, in all positions (figs. 2; 3).2 Regan et al (1991)23 found that resistance to stretch was greatest in the anterior MCL, followed by the posterior MCL, with the RCL being markedly more elastic. However, Morrey and An (1985)24 found that the MCL increased in length more than the RCL, and Kim et al (2002)21 noted only a small increase (1.2 mm) in RCL fibres to the annular ligament between 0o and 90o flexion, although the radial head (to which it is also attached) moved distally by 3.0 mm. These length changes differ from those expected from the stretch resistance values presumably because they were measured in cadavers and thus not a true reflection of joint dynamics in-vivo. Stretch resistance is probably more reliable than positional change in this context as the material properties of these tissues are likely to have developed in response to their functional requirement; dissection and surgery alter tissue mechanics by their very nature.44
In addition, the tendency for the annular ligament to move proximally (with the radial head) during extension was found to be resisted by distal fibres of the LUCL/accessory ligament which did not change in length; but if the LUCL were incomplete, as commonly found in certain groups, an alternative means of stabilizing the annular ligament would be required.21
The tensioned network
 A stout fascial band from the origin of extensor carpi ulnaris consistently inserts on the ulna at the supinator crest (fig. 3) and merges with the LUCL, running just posterior to it, and other connective tissue layers;21,22,37this provides an aponeurotic origin for supinator (figs. 4; 5) which is also attached to the annular ligament and RCL.2,22 These ligaments could be considered as integrated parts of the connective tissue apparatus through which supinator transfers its tension rather than distinct entities.45
A stout fascial band from the origin of extensor carpi ulnaris consistently inserts on the ulna at the supinator crest (fig. 3) and merges with the LUCL, running just posterior to it, and other connective tissue layers;21,22,37this provides an aponeurotic origin for supinator (figs. 4; 5) which is also attached to the annular ligament and RCL.2,22 These ligaments could be considered as integrated parts of the connective tissue apparatus through which supinator transfers its tension rather than distinct entities.45
Traditionally, the fascial connective tissues surrounding muscle are removed during dissection on the implicit assumption that their influence is negligible. Using novel techniques Van der Wal (2009)45 showed that the architecture involved in the transmission of tension in the elbow differs substantially from that described in most textbooks.2 In the lateral elbow region the extensor muscles take origin in an oblique or pennate configuration within strong intermuscular connective tissue layers that converge on the lateral epicondyle as the common extensor tendon (fig. 5).2 These layers form muscular compartments which are also attached to the lateral ulna and radius and are continuous with the antebrachial fascia. A dissection that removed muscle fibres rather than connective tissue revealed that the RCL, LUCL and annular ligaments could not be distinguished as separate entities and were artefacts of connective tissue dissection. A similar situation was found with the MCL and muscles originating from the medial elbow.45
In addition, the orientation of most collagenous fibres in the elbow was found to occur between skeletal tissue and muscle fascicles rather than bone to bone as would be expected with a passive restraining ligament. With the exception of anconeus and part of extensor carpi radialis longus no other muscle attaches directly to periosteum in this region. Transmission of tension is thus primarily through muscles acting in series with collagenous connective tissues, conventionally distinguished as ligaments, aponeuroses, fascial bands etc, and all form part of an interconnected unit.45-48
Using muscles with associated fascia intact, Huijing et al (2003)47 showed that the forces appearing at both ends of a stimulated muscle differed and were dependent on muscle length and the relative positions of adjacent muscles and associated fascia. As ‘deep’ fascia covers all the muscles as the brachial and antebrachial fascia, and receives fibrous expansions from most of them,2,37,38 it directly affects the amount of tension produced by synergist47 and even antagonist muscles.48 Because length changes of poly-articular muscles are determined across two or more joints they are more likely to change their relative positions with respect to mono-articular muscles and alter the tension in associated fascia as the joint angle changes. Proximally these fasciae are continuous with the axillary, pectoral and deltoid fasciae and distally with the palmar fascia, thus contraction of almost any muscle is likely to create tension in these myofascial sheets, influencing joint dynamics locally and at a distance.37,49 Mechanical isolation of anconeus from the fascial system would enable it to regulate joint spacing/pressure through neural signalling alone.37,45
Dynamic stabilization of the elbow joint through muscle action is not a new idea9,16,21,22,50 but to consider ligaments as acting independently, in a piecemeal like way, is a view that is no longer tenable. Importantly, their inclusion into a single architectural system lends support to the tensegrity concept and the proposed functions for brachioradialis and anconeus.
Proprioception and neural control
As myofascial connections create anatomical continuity between different muscles they allow reciprocal feedback over multiple pathways mechanically and neurally.38,51The attachment of muscle spindles to the perimysium suggests that they are monitoring connective tissue tension rather than muscle activity per se49 and a variety of both free and encapsulated nerve endings occur within the brachial and antebrachial fascia.52-55
Proprioceptive receptors monitor the precise timing, intensity, duration and release of tissue deformation while free nerve endings are mostly nociceptive. Encapsulated Pacinian corpuscles and Golgi organs react to local tissue compression due to rapid movement and vibration, while partially-encapsulated Ruffini endings respond to changes in axial tension and provide long lasting information relative to deformation that is slowly adaptive;52,56 they would all be expected to concentrate in transitional areas between tissues with differing mechanical properties. Indeed, all these sensors were found to be densely distributed near proximal and distal attachments of the anterior and posterior MCL, and evenly distributed in the oblique MCL.56 In the RCL, Golgi organs, Ruffini endings and free nerve endings were found to be present, but not Pacinian, with their density greatest in the distal part;56 all were evenly distributed in the ‘annular ligament’.27 Van der Wal (2009)45 showed these sensors to be organized according to the functional architecture of muscles arranged in series with dense connective tissue layers, such as ligaments and aponeuroses, challenging the usual distinction between muscular and articular mechanoreceptors and supporting the findings described earlier.
Ligamentous tissues and regions of the capsule that become stretched during any particular movement, and generate proprioceptive information, are supplied by the same nerve that innervates muscles capable of removing that stress, according to Hiltons law;2 they influence muscle activity through interconnections in the spinal cord and supraspinal centres.57 Linking brachioradialis, anconeus and the posterior capsule through the same nerve supply (radial) makes functional sense if they are part of a rapid response unit that regulates pressure across the joint.
If such ‘opposing’ muscles work as a co-operative synergy to control any particular movement in the elbow the whole concept of flexor/extensor or agonist/antagonist is thus questionable.33,40,51,57,58 Garcia-Ellis et al (2008) proposed that brachioradialis also works co-operatively with biceps to control pressures exerted across the distal radio-ulnar joint.59 Because any component within a synergy can have multiple roles in relation to other synergies, its ability to function within each of them may depend on a huge number of mechanical and neural interactions within the system.51,57 Considering muscles as ‘dynamic ligaments’ that regulate tension through connective tissues within a tensegrity system provides a global mechanism for controlling joint surfaces during movement.13,45,51
DISCUSSION
Research
Most experimental work that has considered biological tensegrity has been carried out on cells,60-64 which are essentially complete organisms that can be examined individually, collectively, or in parts. Single cells are easily manipulated, abundant in variation and supply, and there are few issues that might restrict their use; multi-celled organisms such as humans, however, are quite different. They consist of multiple hierarchical levels with all the increases in complexity that might be expected and examination of any part in isolation can be misleading.44 The experimental use of cadavers has its limitations because they do not give results that exactly match living structures and in vivo experimentation is restricted due to practical and ethical issues. Generalizations of tensegrity from a macro perspective have thus been reasoned from first principles or inferred from models and observation.13-15,51,60,65
Engineers have successfully applied the tensegrity concept to robotics and space research, and progressed understanding of the underlying mathematics,66-68 but have also introduced a classification that is questionable from a biological perspective: class 1 – where compression struts do not touch, as defined by Snelson6 and Fuller,7 and class 2 – where the struts are connected directly to each other.66 This division might be useful in engineering but is doubtful when considering joints as hierarchical structures,12-15 i.e. a description of the elbow as a compressed hinge (class 2), such as in cadavers,2,66 could be different in-vivo when considered in the detail described here (class 1). Levin (2002)13 proposed the term ‘biotensegrity’ to distinguish between tensegrity in living systems and the artificiality of man-made structures.
Structural efficiency
Both tensegrity models and biological structures exhibit non-linear visco-elastic properties during movement.66-68 This is an unusual finding in man-made structures, which use materials capable of handling damaging stresses such as bending moments, shear and torque (class 2), and require stronger components to counteract the huge forces generated as they get bigger.69 Biological structures self-assemble and have evolved in ways that avoid these stresses by distributing the load amongst smaller components within a hierarchy;70,71 if they didn’t, they are likely to collapse through material fatigue and that would be the end of it.72 The separation of tension and compression into different components means that material properties can be optimized and stresses dissipated down to the molecular level;70,71,73 a significant reduction in mass is achieved in a hierarchical tensegrity arrangement66 and in the natural world the most energy-efficient system always wins out.13
Because tension always acts in straight lines and reduces itself to a minimum, tensegrity structures are geodesics by their very nature (their connections follow the shortest path between two points). Geodesic geometry is fundamental to biological self-assembly because of its close-packing and minimal-energy efficiency.7,13,65 It is also the basis for the helix, an archetypal pattern found in biological tissues and described in terms of tensegrity;7,74 the microstructure of myofascial sheaths75 and helical motion of synovial joints,2,16 including the elbow, are both examples of this.
The cytoskeleton
Ingber (2008)60 described tensegrity in the cellular cytoskeleton as a ‘dynamic information processing network’ that integrates mechanical with chemical signals and alters cell function.61 Cytoskeletal tension also transmits to the extra-cellular matrix through integrins in the cell membrane62,63 and is likely to influence other cells some distance away leading to the emergence of complex multi-cellular tissue patterns.64Growth-generated strains and pressures within developing tissues then mechanically feed back and influence morphogenesis of the entire limb.76 It has been proposed that this is part of a tensegrity hierarchy that extends from the molecular level through the cellular and multi-tissue domains to link the entire limb and body.12-14,52,65 Considering the elbow from a tensegrity perspective, where the basic forces of tension and compression are linked mechanically and neurally, provides a view of the living structure that is compatible with the evidence.
CONCLUSION
The idea of a joint functioning as a unit because of global tension rather than piecemeal activity of its components has been around for some time9 but it is only recently that the anatomical details supporting multiple tension pathways have been recognized.37,38,45-49 The essence of a tensegrity system is that every component contributes to structural stability throughout movement, rather than a ‘flexor’ pulling one way, ‘extensor’ the other, and ligaments limiting motion. Numerous muscular expansions into the fascia and smooth muscle cells within it maintain a constant basal pre-tension in-vivo even at rest,37,53 a pre-requisite to tensegrity and one of the reasons why cadaver experiments have their limitations. As tension always reduces itself to a minimum and equilibrates throughout the system, reciprocal interactions between muscles and connective tissues, if arranged in a tensegrity configuration, would provide the most energy efficient means of moving the joint and maintain stiffness and stability.11-14,66,67 This outsourcing of complex control tasks directly into the structure reduces the effort needed to coordinate changes49,77,78 with muscles refining movement at a higher level of control. It may be that variations in muscle and fascial connections between individuals are structural alternatives that efficiently reduce unnecessary redundancy during early development
Tensegrity is a complete and inclusive structural system, and the elbow as such would mechanically rely on the whole limb in order to function normally, which in turn would depend on the entire body interacting in a reciprocal way. Postural abnormalities, misuse and injury in tissues some distance away could affect structures around the elbow, causing changes in local tensional balance and paradoxical proprioceptive activity during movement.49,54,55 A neurally mediated response through the muscle system may attempt to compensate for this but connective tissues take longer to adapt and are relatively passive;52,53 consequently, dynamic stability of the joint and tensegrity system could be compromised, at least temporarily. Unless this is resolved, nociceptive signalling is likely to increase under certain loading conditions and may give rise to the symptoms of such vague pathologies as ‘tennis elbow’ and ‘repetitive strain injury’. If this situation continues, the normal relationship between joint surfaces and tissues under tension might change irreversibly, ultimately leading to structural changes in soft tissues, bone and cartilage that characterise ‘osteoarthrosis’.
If the elbow is part of a global tensegrity configuration, each component will be contributing to its function throughout normal movement,13,51 and any ‘aberrant’ mechanical or neural signal has the potential to compromise it. The tensegrity concept is compatible with the view of the body as a complex mechanical unit and the principles of manual treatment used to restore normality to the elbow and other joints.
REFERENCES
- Gerbeux M, Turpin E, Lensel-Corbeil G. Musculo-articular modelling of the triceps brachii. J Biomech 29:171-80.
- Standring S. Gray’s Anatomy. 39th ed. Elsevier Churchill Livingstone; 2005.
- Pough FH, Janis CM, Heiser JB. Vertebrate life. Prentice Hall; 2002.
- Andersson K, Werdelin L. The evolution of cursorial carnivores in the tertiary: implication of elbow-joint morphology. Proc R Soc Lond B 2003;270:S163-5.
- Fa-Hong Y, Yan-Zhang P, Ru-Liang P, Zhi-Zhang Y, Hong W, Jablonski NG. A comparative study of the elbow joints in five species of Chinese macaques. Folia Primatol 1993;60:18-27.
- Heartney E. Kenneth Snelson: forces made visible. Hard Press Editions; 2009.
- Fuller BB. Synergetics, Explorations in the Geometry of Thinking. Macmillan; 1975.
- Ingber DE, Madri JA. Jamieson JD. Role of basal lamina in neoplastic disorganization of tissue architecture. Proc Nat Acad Sci 1981;78:3901-5.
- Levin SM. Continuous tension, discontinuous compression: a model for biomechanical support of the body. Bull Struct Integ 1982;8.
- Lighthill J. The recently recognized failure of predictability in Newtonian dynamics. Proc R Soc Lond: A 1986;407:35-50.
- Levin SM.Unpublished. See: http://www.biotensegrity.com/in_vivo_observation_of_knee_joints.php
- Levin SM. Putting the shoulder to the wheel: a new biomechanical model for the shoulder girdle. J Biomed Sci Instrum 1997;33:412-7.
- Levin SM. The tensegrity truss as a model for spine mechanics: biotensegrity. J Mech Med Biol 2002;2:375-88.
- Levin SM. A suspensory system for the sacrum in pelvic mechanics: biotensegrity. In: Vleeming A, Mooney V, Stoeckart R., editors. Movement, Stability and Lumbopelvic Pain. Churchill Livingstone; 2007.
- Scarr GM. A model of the cranial vault as a tensegrity structure, and its significance to normal and abnormal cranial development. Int J Osteo Med 2008:11:80-9.
- Hagert E, Hagert CG. Understanding stability of the distal radioulnar joint through an understanding of its anatomy. Hand Clin 2010;26:459-66.
- Rikli DA, Honigmann P, Babst R, Cristalli A, Morlock MM, Mittlmeier T. Intra-articular pressure measurement in the radioulnocarpal joint using a novel sensor: in vitro and in vivo results. J Hand Surg Am 2007;32:67-75.
- Marai GE, Crisco JJ, Laidlaw DH. A kinematics-based method for generating cartilage maps and deformations in the multi-articulating wrist joint from CT images. Proc 28th IEEE EMBS Annual Internat Conf 2006:2079-82.
- Baeyens JP, Glabbeek FV, Goossens M, Gielen J, Van Roy P, Clarys JP. In vivo 3D arthrokinematics of the proximal and distal radioulnar joints during active pronation and supination. Clinical Biomechanics 2006; 21:S9-12.
- Martin BF. The annular ligament of the superior radio-ulnar joint. J Anat 1958;52:473-82.
- Kim P, Isogai S, Murakami G, Wada T, Aoki M, Yamashita T, et al. The lateral collateral ligament complex and related muscles act as a dynamic stabilizer as well as a static supporting structure at the elbow joint: an anatomical and experimental study. Okajimas Folia Anat Jpn 2002;79:55-62.
- Cohen MS, Hastings H. Rotatory instability of the elbow. J Bone Joint Surg 1997;79A:225-33.
- Regan WD, Korinek SL, Morrey BF, An K. Biomechanical study of ligaments around the elbow joint.Clin Orthop Rel Res 1991;271:170-9.
- Morrey BF, An K. Functional anatomy of the ligaments of the elbow. Clin Orthop Rel Res 1985;201:84-90.
- Eckstein F, Lohe F, Hillebrand S, Bergmann M, Schulte E, Milz S, et al. Morphomechanics of the humero-ulnar joint: I. joint space width and contact areas as a function of load and flexion angle. Anat Rec 1995;243:318-26.
- Eckstein F, Merz B, Muller-Gerbl M, Holzknecht N, Pleier M, Putz R. Morphomechanics of the humero-ulnar joint: II. Concave incongruity determines the distribution of load and subchondral mineralization. Anat Rec 1995;243:327-35.
- Levangie PK, Norkin CC. Joint structure and function – a comprehensive analysis. 3rd ed., FA Davis, Philadelphia 2001.
- Stroyan M, Wilk KE. The functional anatomy of the elbow complex. JOSPT 1993;17:279-88.
- Skahen JR, Palmer AK, Werner FW, Fortino MD. The interosseous membrane of the forearm: anatomy and function. J Hand Surg 1997;22:981-5.
- Sircar S. Principles of medical physiology. Georg Thieme Verlag; 2008.
- Johnson DL, Gellman H, Waters RL, Tognelia M. Brachioradialis transfer for wrist extension in tetraplegic patients who have fifth-cervical-level neurological function. J Bone Joint Surg 1996;78:1063-7.
- Ozkan T, Tuncer S, Aydin A. Brachioradialis re-routing for the restoration of active supination and correction of forearm pronation deformity in cerebral palsy. J Hand Surg Br 2004;29B:265-70.
- Pauly JE, Rushing JL, Scheving LE. An electromyographic study of some muscles crossing the elbow joint. Anat Rec 2005;159:47-54.
- Le Bozec S, Maton B, Cnockaert JC. The synergy of elbow extensor muscles during static work in man. Eur J Appl Physiol 1980;43:57-68.
- Gleason TF, Goldstein WM, Ray RD. The function of the anconeus muscle. Clin Orthop relat Res 1985;192:147-8.
- Basmajian JV, Griffin WR. Function of anconeus muscle. J Bone Joint Surg Am 1972;54:1712-4.
- Stecco C, Gagey O, Macchi V, Porzionato A, de Caro R, Aldegheri R, et al. Tendinous muscular insertions onto the deep fascia of the upper limb. First part: anatomical study. Morphologie 2007;91:29-37.
- Stecco A, Macchi V, Stecco C, Porzionato A, Day JA, Delmas V, et al. Anatomical study of myofascial continuity in the anterior region of the upper limb. J Bodyw Mov Ther 2009;13:53-62.
- Nakazawa K, Kawakami Y, Fukunaga T, Yano H, Miyashita M. Differences in activation patterns in elbow flexor muscles during isometric, concentric and eccentric contractions. Eur J Appl Physiol 1993;66:214-20.
- Naito A, Sun Y, Yajima M, Fukamachi H, Ushikoshi K. Electromyographic study of the elbow flexors and extensors in a motion of forearm pronation/supination while maintaining elbow flexion in humans. Tohoku J Exp Med 1998;186:267-77.
- Jacobs CR, Eckstein F. Computer simulation of subchondral bone adaptation to mechanical loading in an incongruous joint. Anat Rec 1997;249:317-26.
- Milz S, Eckstein F, Putz R. Thickness distribution of the subchondral mineralization zone of the trochlear notch and its correlation with the cartilage thickness: an expression of functional adaptation to mechanical stress acting on the humeroulnar joint? Anat Rec 1997;248:189-97.
- Sverdlova NS, Witzel U. Principles of determination and verification of muscle forces in the human musculoskeletal system: muscle forces to minimise bending stress. J Biomech 2010;43:387-96.
- Moore SM, Ellis B, Weiss JA, McMahon PJ, Debski RE. The glenohumeral capsule should be evaluated as a sheet of fibrous tissue: a validated finite element model. Annals Biomed Eng 2010;38:66-76.
- Van der Wal J. The architecture of the connective tissue in the musculoskeletal system – an often overlooked functional parameter as to proprioception in the locomotor apparatus. Int J Thera Mass Bodyw 2009;2:9-23.
- Maas H, Jaspers RT, Baan GC, Huijing PA. Myofascial force transmission between a single muscle head and adjacent tissues: length effects of head III of rat EDL. J Appl Physiol 2003;95:2004-13.
- Huijing PA, Baan GC. Myofascial force transmission: muscle relative position and length determine agonist and synergist muscle force. J Appl Physiol 2003;94:1092-107.
- Huijing PA, Baan GC. Myofascial force transmission via extramuscular pathways occurs between antagonistic muscles. Cells Tiss Org 2008;188:400-14.
- Stecco L. Fascial manipulation for musculoskeletal pain. Piccin Nuova Libraria; Padova; 2004.
- Gibbons SGT, Mottram SL, Comerford MJ. Stability and movement dysfunction related to the elbow and forearm. Orthop Div Rev 2001;Sept/Oct:15–33.
- Turvey MT. Action and perception at the level of synergies. Hum Mov Sci 2007;26:657-97.
- Schleip R. Fascial plasticity – a new neurobiological explanation: part 1. J Bodyw Mov Ther 2003;7:11-9.
- Schleip R. Fascial plasticity – a new neurobiological explanation: part 2. J Bodyw Mov Ther 2003;7:104-16.
- Stecco C, Gagey O, Belloni A, Pozzuoli A, Porzionato A, Macchi V, et al. Anatomy of the deep fascia of the upper limb. Second part: study of innervation. Morphologie 2007;91:38-43.
- Stecco C, Porzionato A, Macchi V, Tiengo C, Parenti A, Aldegheri R, et al. A histological study of the deep fascia of the upper limb. <i”>It J Anat Embryol 2006;111:125-32.</i”>
- Petrie S, Collins JG, Solomonow M, Wink C, Chuinard R, D’Ambrosia R. Mechanoreceptors in the human elbow ligaments. J Hand Surg 1998;23A:512-8.
- Bawa P, Chalmers GR, Jones KE, Sogaard K, Walsh ML. Control of the wrist joint in humans. Eur J Appl Physiol 2000;83:116-27.
- Sella GE. Elbow muscular relationships: an SEMG view of muscular agonism and antagonism. J Neurol Orthop Med Surg 2003;21:1-10.
- Garcia-Ellis M, Lluch AL, Ferreres A, Lluch A, Lhamby F. Transverse loaded pronosupination test. J Hand Surg Eur 2008;33E:765-7.
- Ingber DE. Tensegrity-based mechanosensing from macro to micro. [Review] Biophys Mol Biol 2008;9:163-79
- Stamenovic D, Ingber DE, Tensegrity-guided self assembly: from molecules to living cells. Soft Matt 2009;5:1137-45. Review
- Vogel V, Sheetz M. Local force and geometry sensing regulate cell functions. Nature Rev Mol Cell Biol 2006;7:265-75.
- Wang N, Tytell JD, IngberDE. Mechanotransduction at a distance: mechanically coupling the extracellular matrix with the nucleus. Nature Rev: Mol Cell Biol 2009;10:75-82.
- Nelson CM, Jean RP, Tan JL, Liu WF, Sniadecki NJ, Spector AA, et al. Emergent patterns of growth controlled by multicellular form and mechanics. Proc Nat Acad Sci 2005;102:11594-9.
- Scarr G. Simple geometry in complex organisms. J Bodyw Mov Ther 2010;14:424-44.
- Skelton RE, Adhikari R, Pinaud JP, Chan W, Helton JW. An introduction to the mechanics of tensegrity structures. Proc. 40th IEEE Conf. Decision and Contr 2001;5
- Masic M, Skelton RE, Gill PE Optimization of tensegrity structures. Inter J Solids
- Juan SH, Tur JMM. Tensegrity frameworks: static analysis review. Mechanism Machine Theory 2008;43:859-81.
- Gordon JE. Structures or why things don’t fall down. Penguin; 1978.
- Gao H, Ji B, Jager IL, Arzt E, Fratz P. Materials become insensitive to flaws at nanoscale: lessons from nature. Proc Nat Acad Sci 2003;100:5597-600.
- Gupta HS, Seto J, Wagermaier W, Zaslansky P, Boesecke P, Fratzl P. Cooperative deformation of mineral and collagen in bone at the nanoscale. Proc Nat Acad Sci 2006;103:17741-46.
- Salvadori M. Why buildings stand up: the strength of architecture. Norton; 2002, p.83.
- Zanotti G, Guerra C. Is tensegrity a unifying concept of protein folds? FEBS Letters 2003;534:7-10.
- Scarr G. Helical tensegrity as a structural mechanism in human anatomy. Int J Osteo Med 2011;14:24-32.
- Purslow PP. Muscle fascia and force transmission. J Bodyw Mov Ther 2010;14:411-7.
- Henderson JH, Carter DR. Mechanical induction in limb morphogenesis: the role of growth-generated strains and pressures. Bone 2002;31:645-53.
- Valero-Cuevas FJ, Yi JW, Brown D, McNamara RV, Paul C, Lipson H. The tendon network of the fingers performs anatomical computation at a macroscopic scale. Trans Biomed Eng 2007;54:1161-66.
- Rieffel JA, Valero-Cuevas FJ, Lipson H. Morphological communication: exploiting coupled dynamics in a complex mechanical structure to achieve locomotion. J R Soc: Interface 2010;7:613-21.
